Sanofi’s Dengvaxia was a deadly shipwreck in the Philippines. Takeda recently published preliminary results of its new vaccine—quite careful in projecting impact. Rightfully so.
For the moment, Takeda reported preliminary results that are decidedly mixed. When taking into account all four serotypes, or strains, of the dengue virus, the Takeda vaccine demonstrated an overall 80.2% effectiveness rate. The late-stage study, published in the New England Journal of Medicine, tested two doses in more than 19,000 children and adolescents in eight countries.
Breaking down the results further, though, shows a more nuanced story. In one strain — known as DENV-2 — the effectiveness rate was an impressive 97.7%. But effectiveness in two other strains — DENV-1 and DENV-3 — was much lower, at 73.7% an 62.6%, respectively. There was not enough data to assess the extent to which the vaccine mitigated the fourth strain, known as DENV-4.
“We’re very encouraged by the results,” Derek Wallace, a Takeda vice president who heads the dengue vaccine project, told us. Despite the moderate effectiveness displayed in two of the strains, the data “still represents a massive step forward in the reduction of the burden of the disease. We see it encouragingly when you consider the burden of (the) dengue (virus) globally.”
“We’re looking very carefully at the safety of this vaccine candidate on the assumption it continues to have no important safety side effects. A vaccine which has partial efficacy against some serotypes would still have significant public health advantages,” he continued, noting more data will be released later this month at a scientific conference. Wallace added that efficacy against hospitalization was also high.
i.e. w – a – i – t.
+
Death (% and #) trends due to Dengue in the following select countries versus globally:
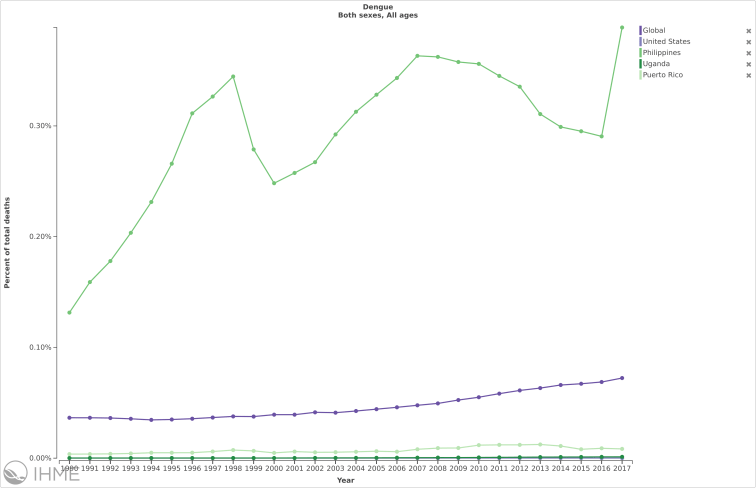
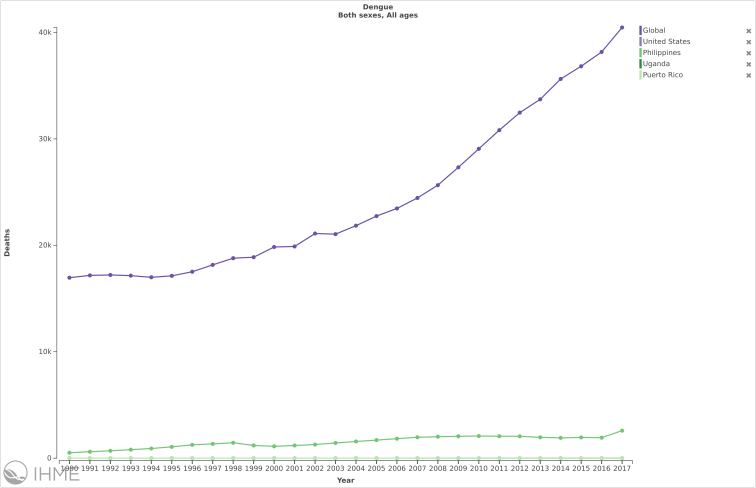
Source: IHME/GBD 2017/University of Washington

You must be logged in to post a comment.